Emissions of CO2 increasing ocean acidification with future serious risks to marine life
The world’s oceans are becoming acidic at an “unprecedented rate” and this may be happening at faster than at any time in the past 300 million years. In their strongest statement yet on this issue, a large number of scientists say ocean acidification could increase by 170% by 2100. They say that some 30% of ocean species are unlikely to survive in these conditions. The researchers conclude that human emissions of CO2 are clearly to blame – humanity is putting some 24 million tonnes of CO2 into the oceans each day. That is already altering the chemistry of the waters, and will do so even more in future. Since the start of the industrial revolution, the waters have become 26% more acidic – and there are serious concerns about the impact this is having, and will have, on many ocean species. These include oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton. When shelled organisms are at risk, the entire food web may also be at risk. Today, more than a billion people worldwide rely on food from the ocean as their primary source of protein.
.
Tweet
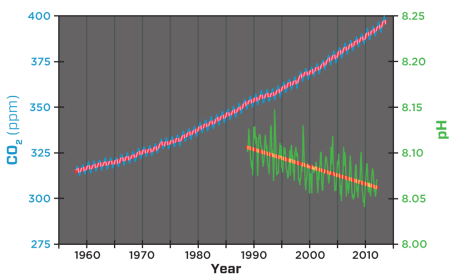
Atmospheric CO2 and ocean pH
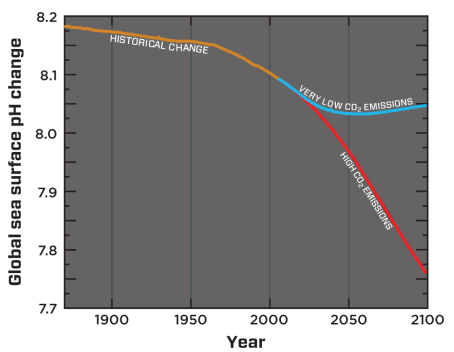
Ocean surface pH projections to 2100
Modelled global sea-surface pH from 1870 to 2100. The blue line reflects estimated pH change resulting from very low CO2 emissions to the atmosphere (IPCC Representative Concentration Pathway, RCP* 2.6). The red line reflects pH from high CO2 emissions (the current emissions trajectory, RCP* 8.5).
Credit: Adapted from Bopp et al., 2013 (reference 9).
Emissions of CO2 driving rapid oceans ‘acid trip’
By Matt McGrath, Environment correspondent,
BBC News
14.11.2013

The world’s oceans are becoming acidic at an “unprecedented rate” and may be souring more rapidly than at any time in the past 300 million years.
In their strongest statement yet on this issue, scientists say acidification could increase by 170% by 2100.
They say that some 30% of ocean species are unlikely to survive in these conditions.
The researchers conclude that human emissions of CO2 are clearly to blame.
The study will be presented at global climate talks in Poland next week.
In 2012, over 500 of the world’s leading experts on ocean acidification gathered in California. Led by the International Biosphere-Geosphere Programme, a review of the state of the science has now been published.
This states with “very high confidence” that increasing acidification is caused by human activities which are adding 24 million tonnes of CO2 to oceans every day.
Pickled waters
The addition of so much carbon has altered the chemistry of the waters.
Since the start of the industrial revolution, the waters have become 26% more acidic.
“This is the state of the art,” said Prof Jean-Pierre Gattuso, from CNRS, the French national research agency.
“My colleagues have not found in the geological record, rates of change that are faster than the ones we see today.”
What worries the scientists is the potential impact on many ocean species including corals.
Studies carried out at deep sea vents where the waters are naturally acidic thanks to CO2, indicate that around 30% of the ocean’s biodiversity may be lost by the end of this century.
These vents may be a “window on the future” according to the researchers.
“You don’t find a mollusc at the pH level expected for 2100, this is really quite a stunning fact,” said Prof Gattuso.
“It’s an imperfect window, only the ocean’s acidity is increasing at these sites, they don’t reflect the warming we will see this century.
“If you combine the two, it could be even more dramatic than what we see at CO2 vents.”
The effect of acidity is currently being felt most profoundly felt in the Arctic and Antarctic oceans. These chilly waters hold more CO2 and increasing levels of the gas are turning them acidic more rapidly than the rest of the world.
The more acidic they become, the more damaging they are to the shells and skeletons of marine organisms.
The researchers say that by 2020, ten percent of the Arctic will be inhospitable to species that build their shells from calcium carbonate. By 2100 the entire Arctic will be a hostile environment.
 Pteropods like this are already feeling the corrosive impact of acidic waters in the Antarctic
Pteropods like this are already feeling the corrosive impact of acidic waters in the AntarcticThese effects are already visible says Prof Gattuso.
“In the Southern Ocean, we already see corrosion of pteropods which are like sea snails, in the ocean we see corrosion of the shell.
“They are a key component in the food chain, they are eaten by fish, birds and whales, so if one element is going then there is a cascading impact on the whole food chain.”
The authors warn that the economic impact of the losses from aquaculture could be huge – the global cost of the decline in molluscs could be $130bn by 2100 if emissions of CO2 continue on their current pathway.
Adding alkaline substances such as crushed limestone to the waters has been mooted as a potential way of mitigating the worst impacts of acidification. But Prof Gattuso says it would only have a limited effect.
“Maybe in bays which have a restricted exchange with open oceans it may work, it may give some local relief.
“But the latest research is showing that it is not really practical at a global scale. It is very expensive and very energy intensive.”
Marine protection zones would also give some short term benefit, but the scientists say that in the long term only significant cuts in emissions will slow the progress of acidification.
http://www.bbc.co.uk/news/science-environment-24904143
.
ACIDIFYING OCEANS
- The oceans are thought to have absorbed up to half of the extra CO2 put into the atmosphere in the industrial age
- This has lowered their pH by 0.1
- pH is the measure of acidity and alkalinity
- It usually ranges from pH 0 (very acidic) to pH 14 (very alkaline); 7 is neutral
- Seawater is mildly alkaline with a “natural” pH of about 8.2
- .
- .
- ..
Related BBC Stories
.
.
Information from NOAA – the American National Oceanic and Atmospheric Administration:
What is Ocean Acidification?
A pH unit is a measure of acidity ranging from 0-14. The lower the value, the more acidic the environment. Becoming more acidic is a relative shift in pH to a lower value.
The Chemistry
When carbon dioxide (CO2) is absorbed by seawater, chemical reactions occur that reduce seawater pH, carbonate ion concentration, and saturation states of biologically important calcium carbonate minerals. These chemical reactions are termed “ocean acidification” or “OA” for short. Calcium carbonate minerals are the building blocks for the skeletons and shells of many marine organisms. In areas where most life now congregates in the ocean, the seawater is supersaturated with respect to calcium carbonate minerals.
This means there are abundant building blocks for calcifying organisms to build their skeletons and shells. However, continued ocean acidification is causing many parts of the ocean to become undersaturated with these minerals, which is likely to affect the ability of some organisms to produce and maintain their shells.
Since the beginning of the Industrial Revolution, the pH of surface ocean waters has fallen by 0.1 pH units. Since the pH scale, like the Richter scale, is logarithmic, this change represents approximately a 30 percent increase in acidity.
Future predictions indicate that the oceans will continue to absorb carbon dioxide and become even more acidic. Estimates of future carbon dioxide levels, based on business as usual emission scenarios, indicate that by the end of this century the surface waters of the ocean could be nearly 150 percent more acidic, resulting in a pH that the oceans haven’t experienced for more than 20 million years.
The Biological Impacts
Ocean acidification is expected to impact ocean species to varying degrees. Photosynthetic algae and seagrasses may benefit from higher CO2 conditions in the ocean, as they require CO2 to live just like plants on land. On the other hand, studies have shown that a more acidic environment has a dramatic effect on some calcifying species, including oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton.
When shelled organisms are at risk, the entire food web may also be at risk. Today, more than a billion people worldwide rely on food from the ocean as their primary source of protein. Many jobs and economies in the U.S. and around the world depend on the fish and shellfish in our oceans.
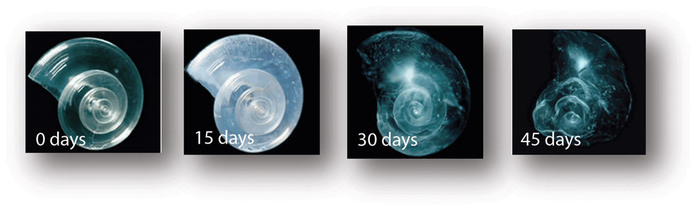
Pteropods
The pteropod, or “sea butterfly”, is a tiny sea creature about the size of a small pea. Pteropods are eaten by organisms ranging in size from tiny krill to whales and are a major food source for North Pacific juvenile salmon.
The photos below show what happens to a pteropod’s shell when placed in sea water with pH and carbonate levels projected for the year 2100. The shell slowly dissolves after 45 days. Photo credit: David Liittschwager/National Geographic Stock. Used with permission. All rights reserved. National Geographic Images.
Shellfish
In recent years, there have been near total failures of developing oysters in both aquaculture facilities and natural ecosystems on the West Coast. These larval oyster failures appear to be correlated with naturally occurring upwelling events that bring low pH waters undersaturated in aragonite as well as other water quality changes to nearshore environments. Lower pH values occur naturally on the West Coast during upwelling events, but a recent observations indicate that anthropogenic CO2 is contributing to seasonal undersaturation. Low pH may be a factor in the current oyster reproductive failure; however, more research is needed to disentangle potential acidification effects from other risk factors, such as episodic freshwater inflow, pathogen increases, or low dissolved oxygen.
It is premature to conclude that acidification is responsible for the recent oyster failures, but acidification is a potential factor in the current crisis to this $100 million a year industry, prompting new collaborations and accelerated research on ocean acidification and potential biological impacts.
Coral
Many marine organisms that produce calcium carbonate shells or skeletons are negatively impacted by increasing CO2levels and decreasing pH in seawater. For example, increasing ocean acidification has been shown to significantly reduce the ability of reef-building corals to produce their skeletons. In a recent paper, coral biologists reported that ocean acidification could compromise the successful fertilization, larval settlement and survivorship of Elkhorn coral, an endangered species. These research results suggest that ocean acidification could severely impact the ability of coral reefs to recover from disturbance. Other research indicates that, by the end of this century, coral reefs may erode faster than they can be rebuilt.
This could compromise the long-term viability of these ecosystems and perhaps impact the estimated one million species that depend on coral reef habitat. For more information on the impact of ocean acidification on coral, see NOAA’s Coral Reef Watch website.
Ocean Acidification: An Emerging Global Problem
Ocean acidification is an emerging global problem. Over the last decade, there has been much focus in the ocean science community on studying the potential impacts of ocean acidification. Since sustained efforts to monitor ocean acidification worldwide are only beginning, it is currently impossible to predict exactly how ocean acidification impacts will cascade throughout the marine food chain and affect the overall structure of marine ecosystems. With the pace of ocean acidification accelerating, scientists, resource managers, and policymakers recognize the urgent need to strengthen the science as a basis for sound decision making and action.
http://www.pmel.noaa.gov/co2/story/What+is+Ocean+Acidification%3F
.
.
.
.
.